Lime Kiln as 7th-Grade Chemistry
We said goodbye to Lucas this morning. He went off to school and won’t return from there until Wednesday evening. His class is in the middle of a chemistry block, and they are staying overnight on the farm to build and mind a lime kiln.
Yes, we totally had to look up “lime kiln.” Thank you, Wikipedia:
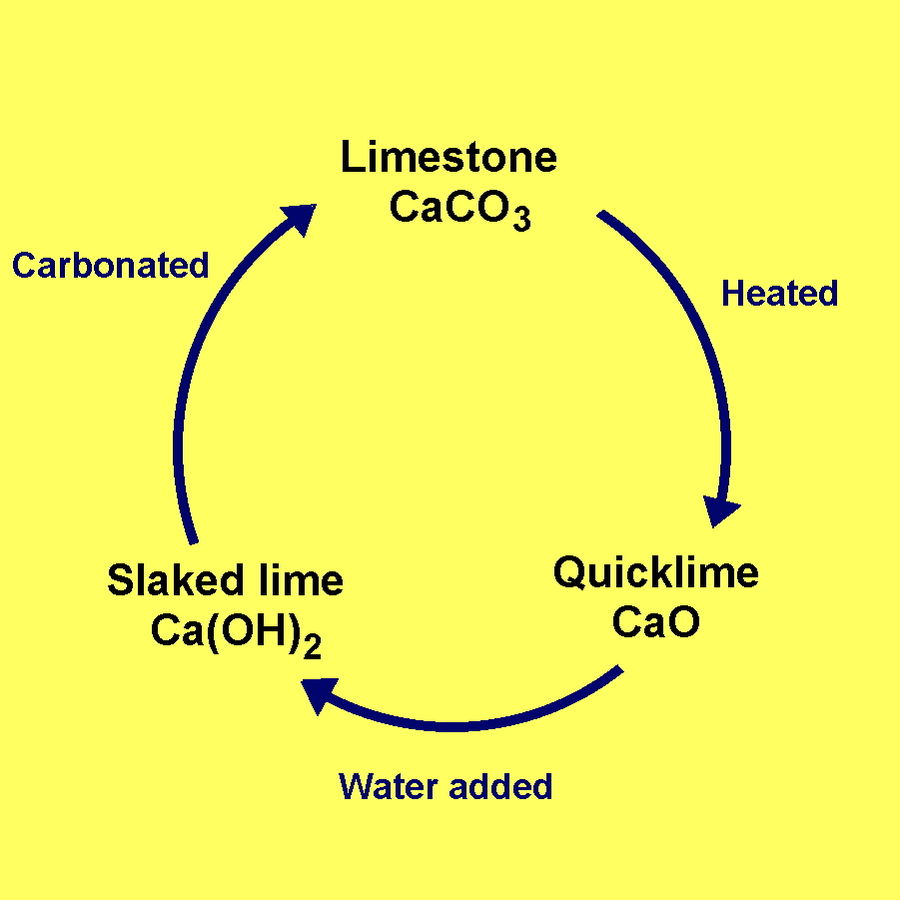
“A lime kiln is used to produce quicklime through the calcination of limestone (calcium carbonate). The chemical equation for this reaction is
CaCO3 + heat → CaO + CO2”
The experience is a recreation of lime plaster, as produced through a series of chemical transformations, known today as the lime cycle. Lime plaster has been used by humans a building material since 5000 BCE.
“Calcium oxide (CaO), commonly known as quicklime or burnt lime, is a widely used chemical compount. It is a white, caustic, alkaline, crystalline solid at room temperature. The broadly used term “lime” connotes calcium-containing inorganic materials, in which carbonates, oxides and hydroxides of calcium, silicon, magnesium, aluminium, and iron predominate. By contrast, “quicklime” specifically applies to the single chemical compound calcium oxide. Calcium oxide which survives processing without reacting in building products such as cement is called free lime.
Quicklime is relatively inexpensive. Both it and a chemical derivative (calcium hydroxide, of which quicklime is the base anhydride) are important commodity chemicals.”
The teacher informed us, “Today’s children have little opportunity to observe actual industrial processes. Almost everything comes magically ready-made and packaged. Nevertheless, the lime cycle studied in 7th Grade chemistry offers an opportunity for the students not only to observe an important industrial process, but to build and fire a kiln used in the process.”
So. SCIENCE! Chemistry. Construction. Fire. Campout at school. Social Arts. Collaboration. 28 seventh graders and three teachers tending a fire through the night in November. Plus a large support staff of parents. I am still amazed at the lengths to which these people will go to give our children a hands-on learning experience.
Now, Lucas is unenthusiastic about this experience. (The current phase of seventh grade seems to involve a lot less enthusiasm for everything. And lots of sighing and rolling of eyes.) He knows it will be cold and hard. He knows he will finish school on Wednesday and have to go straight into the first basketball practice of the season. He knows he will be tired. He won’t be home until dinnertime.
I know he’ll never forget it.